 The principle of conservation of mass was first outlined by Mikhail Lomonosov (1711–1765) in 1748. But if this energy is turned back into mass, you will have the same amount of mass. However, certain nuclear reactions (fusion and fission) can convert a small part of the mass into energy. The law is accurate for all chemical reactions. More simply, whatever you do, you will still have the same amount of stuff.  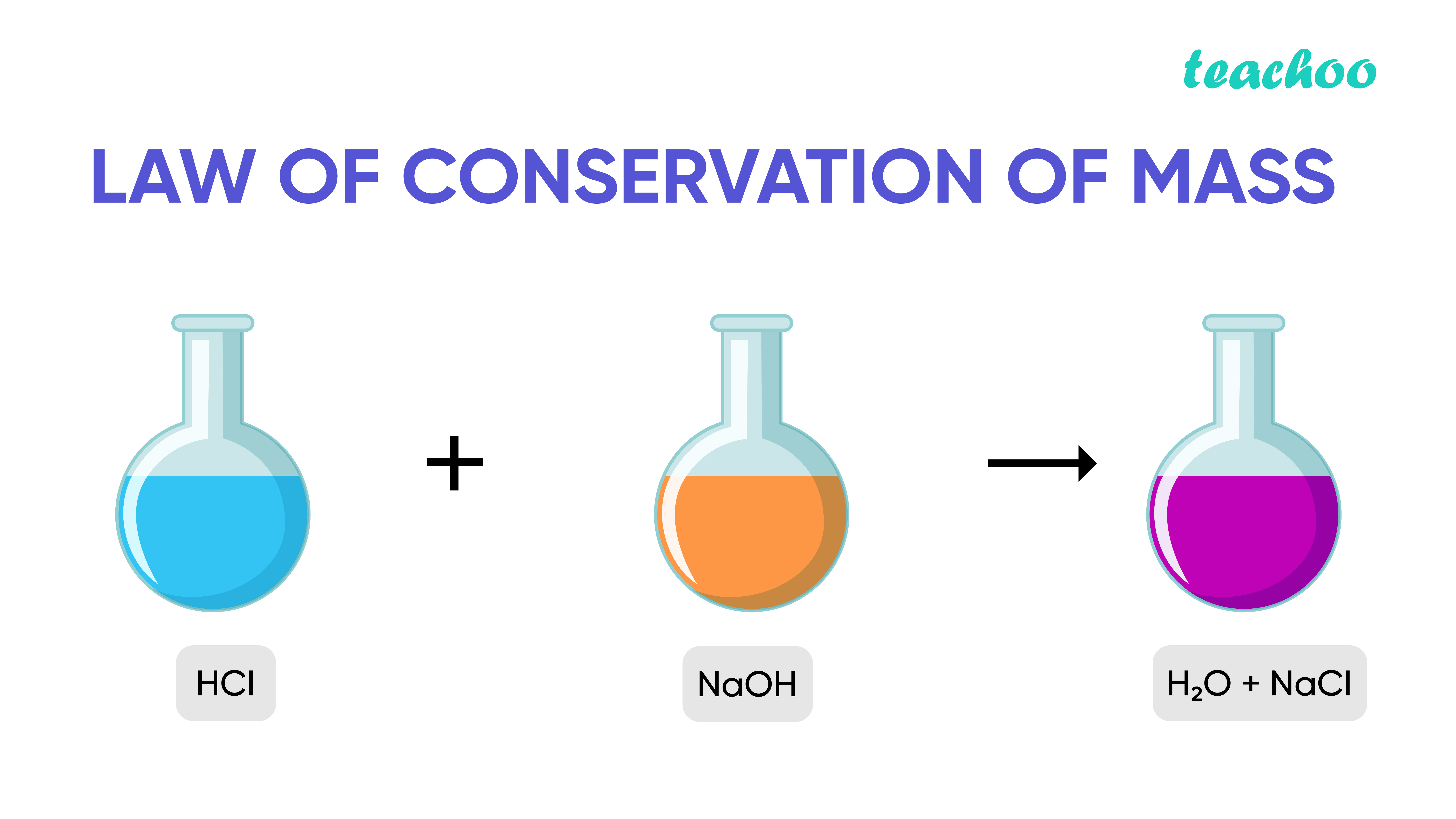
This law says that when a chemical reaction rearranges atoms into a new product, the mass of the reactants ( chemicals before the chemical reaction) is the same as the mass of the products (the new chemicals made). Sir Antoine Lavoisier promoted this idea. Thus, the amount of matter cannot change. 
The law of conservation of mass states that mass can neither be created nor destroyed in a chemical reaction. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
